Procedure Not Recommended
Risks, Complications & Better Alternatives
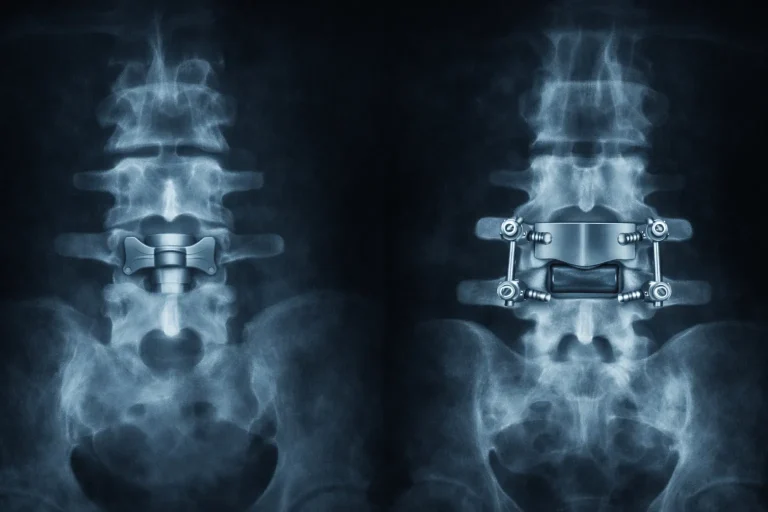
⚠️ While Deuk Spine Institute can perform Interspinous & Interlaminar Device implantation, we do not recommend it.
This page explains why — learn the risks before consenting to this procedure.
UNDERSTANDING THE PROCEDURE
While there are legitimate medical reasons, financial incentives from expensive hardware often influence surgical recommendations.
Nerve root dysfunction from compressed or pinched nerves. However, these devices rarely address the actual source of nerve inflammation.
Severe leg pain from compressed nerves. The device placement destroys stabilizing structures while leaving the herniated disc untreated.
Poly radiculopathy from compressed nerves. A rare emergency often used to justify device implantation for far less severe conditions.
Canal narrowing causing cord compression. Surgeons may overdiagnose this on MRI to justify device implantation for mild cases.
Repeat disc bulging or rupture. Rather than treating the disc itself, surgeons attach hardware that fails to address the root cause.
Each interspinous or interlaminar device generates massive revenue for the manufacturer and hospital, creating a strong incentive to implant.
Surgeons receive premium reimbursement for device implantation procedures, creating powerful financial incentive even when less invasive options exist.
Extended operating time, overnight stays, device costs, and complex post-surgical care drive massive hospital revenue per procedure.
Device migration, separation, and hardware failure are common, guaranteeing additional lucrative revision surgeries and replacements.
Failed device implantations create patients needing ongoing injections, ablations, stimulators, and opioids for life — generating continuous revenue.
Surgeons receive the same compensation whether pain resolves or worsens. The system rewards volume of device implantations, not patient results.
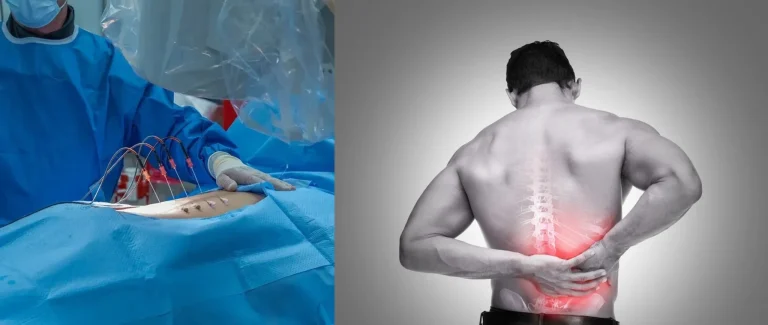
THE SURGICAL PROCESS
The videos below contain real surgical footage. Viewer discretion is advised.
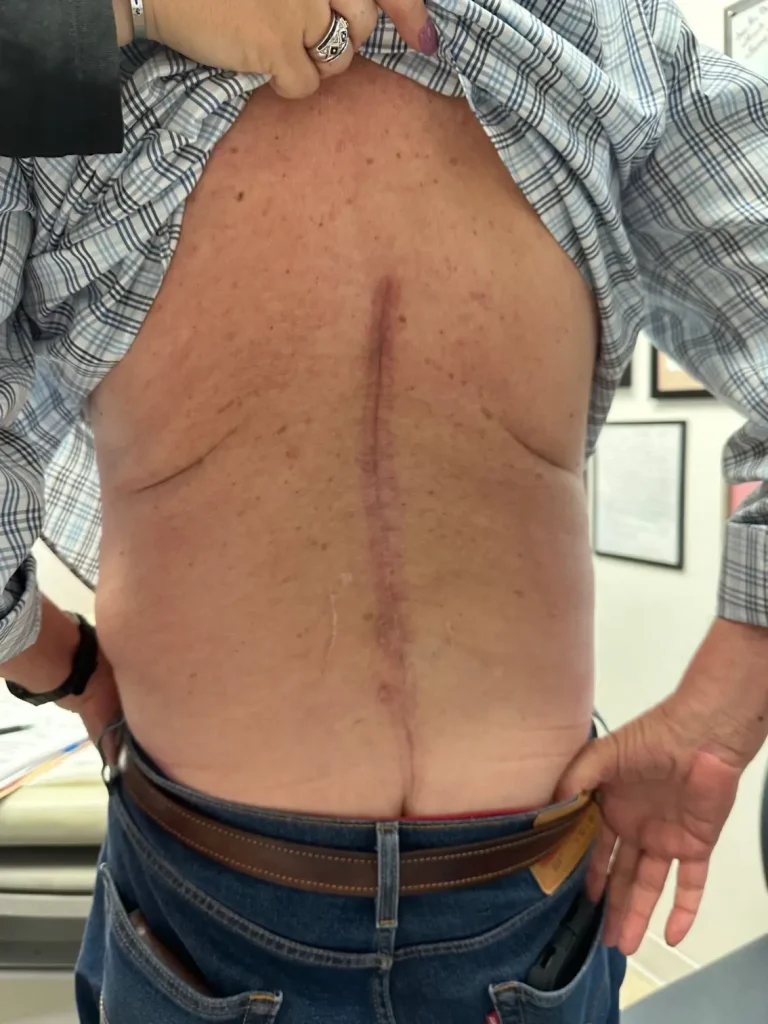
A 2–4 inch incision is made along the spine to expose the underlying vertebral structures.
The tough connective tissue covering the spine muscles is cut and stripped away.
Spinal muscles are stripped from the bone, causing permanent damage and weakness.
The spinous process, interspinous ligaments, and supraspinous ligaments are partially or fully removed.
The lamina and ligamentum flavum are removed, exposing the spinal cord and nerves.
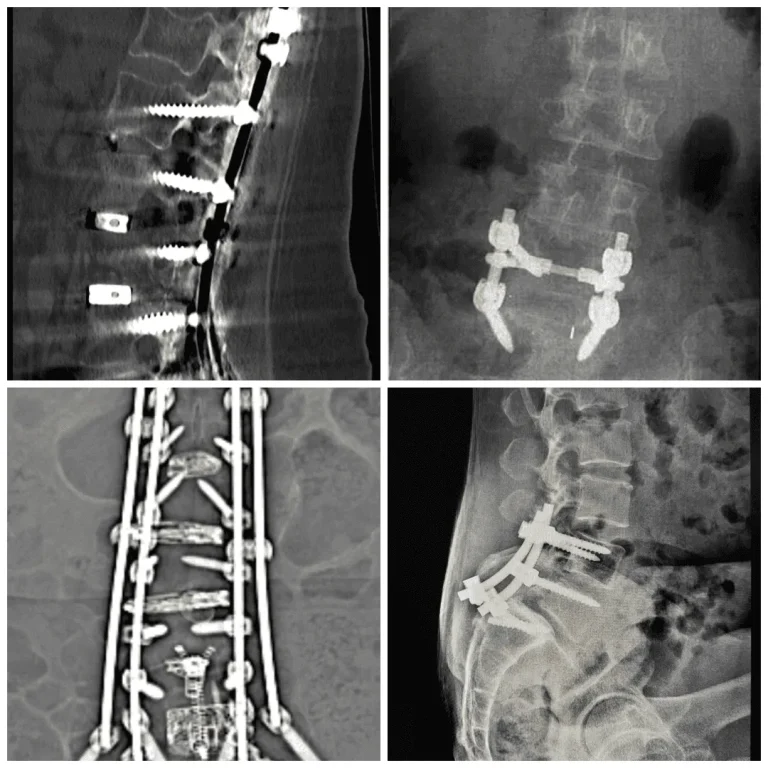
A metal interspinous or interlaminar device is attached between adjacent spinous processes and laminae.
CRITICAL RISKS
15 documented reasons with supporting images and surgical footage.
Images and videos below contain real surgical procedures and medical imagery.
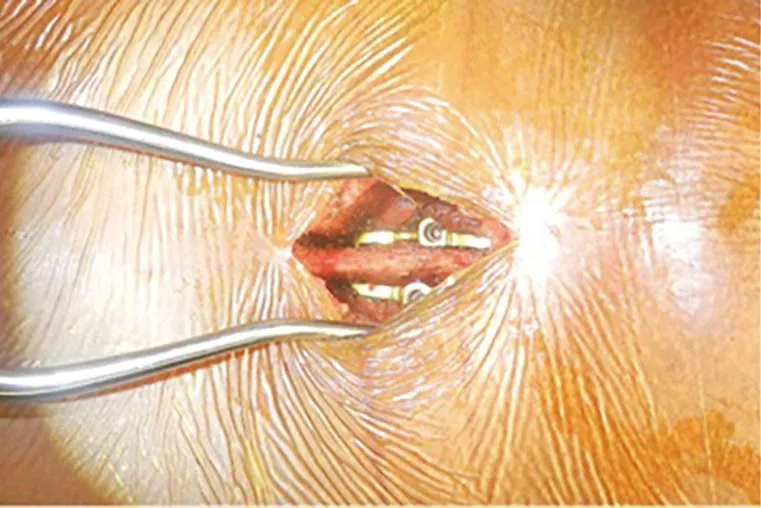

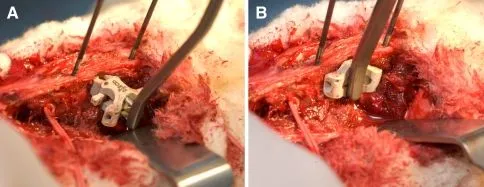

DOCUMENTED COMPLICATIONS
Images and videos below contain real surgical procedures and medical imagery.
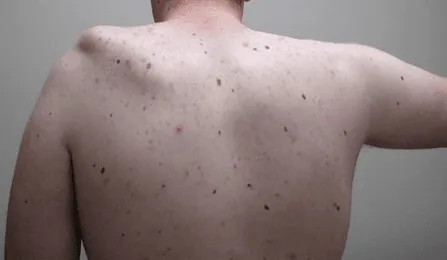
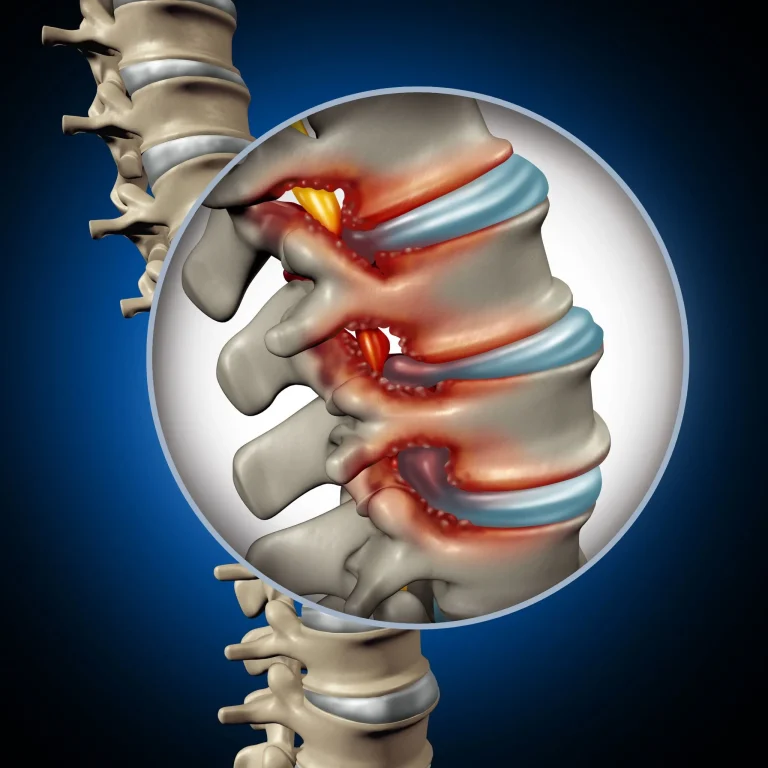
Permanent nerve damage causing weakness, numbness, or paralysis in extremities.
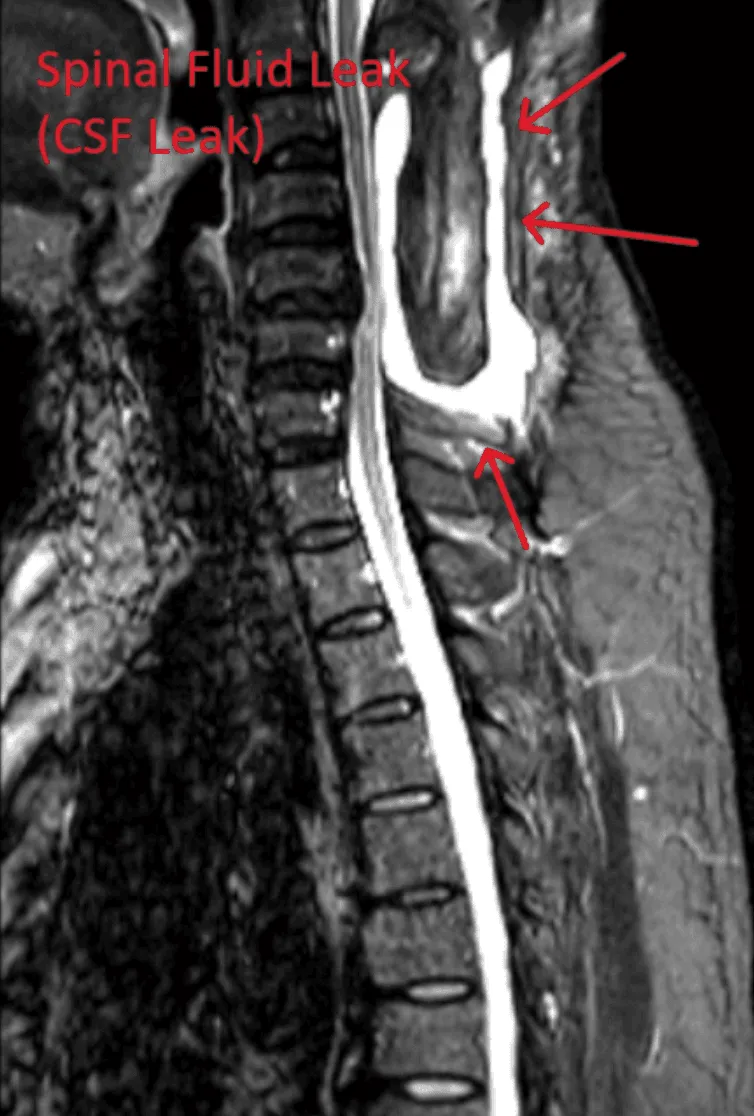
Dural tears leading to cerebrospinal fluid leaks, requiring additional repair surgery.
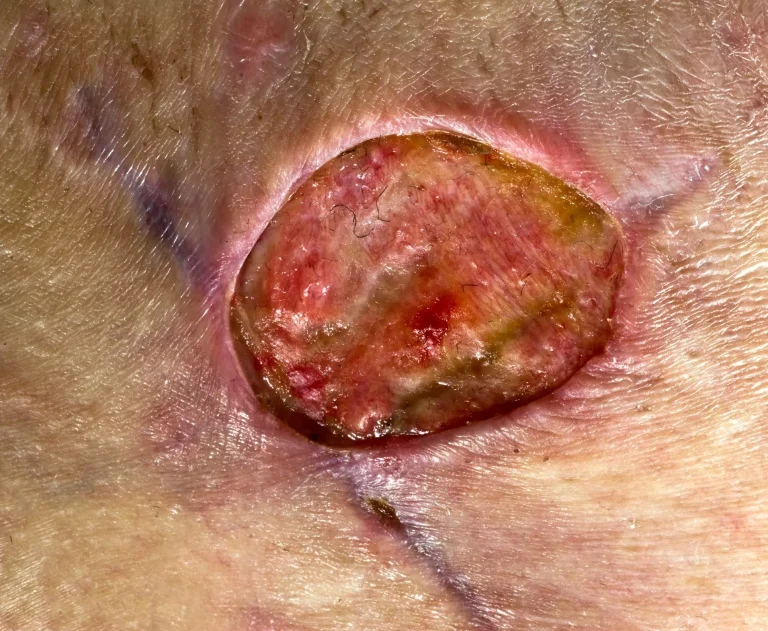
Discitis, osteomyelitis, and infections of blood, spinal fluid, lungs, bladder, and kidneys.
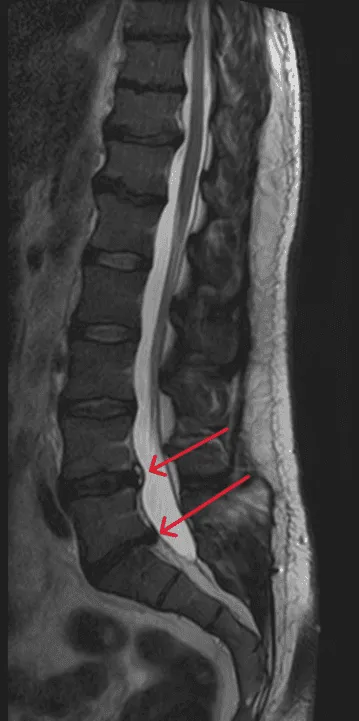
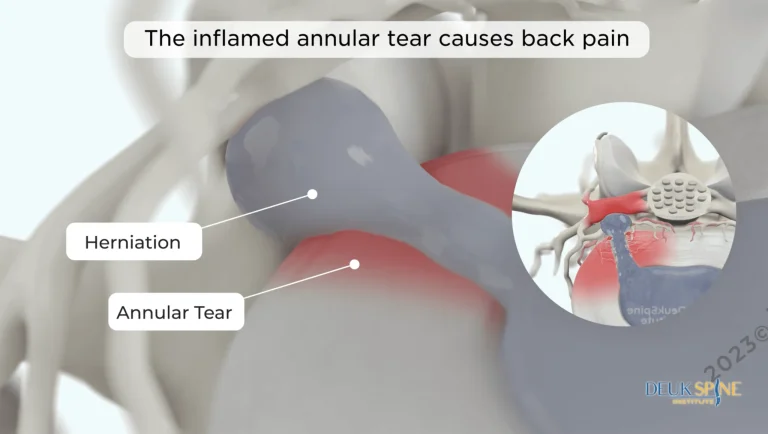
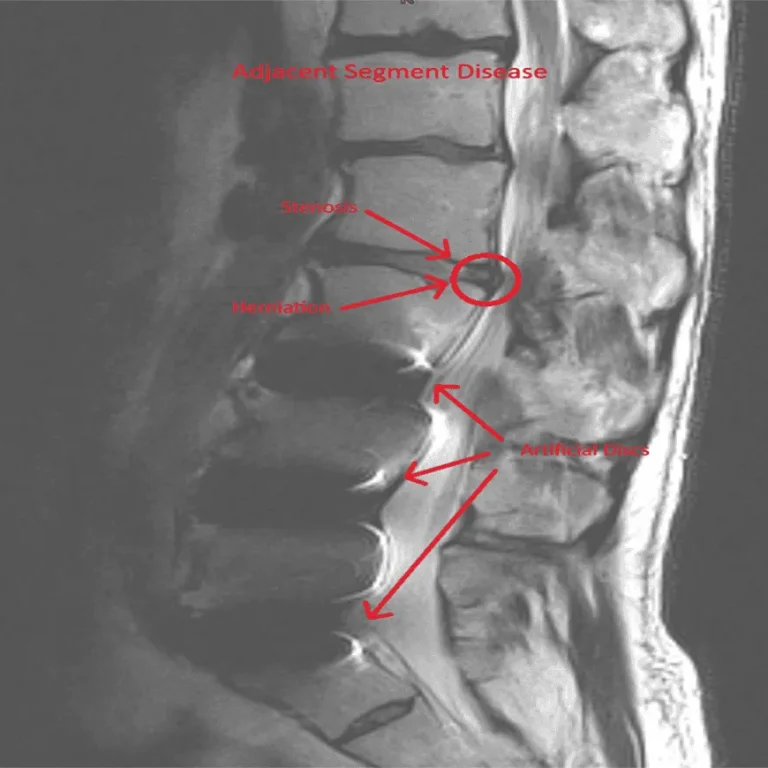
Disc herniation returns because the device does not treat the underlying disc injury.
Stenosis persists or reoccurs after device placement, requiring additional surgery.
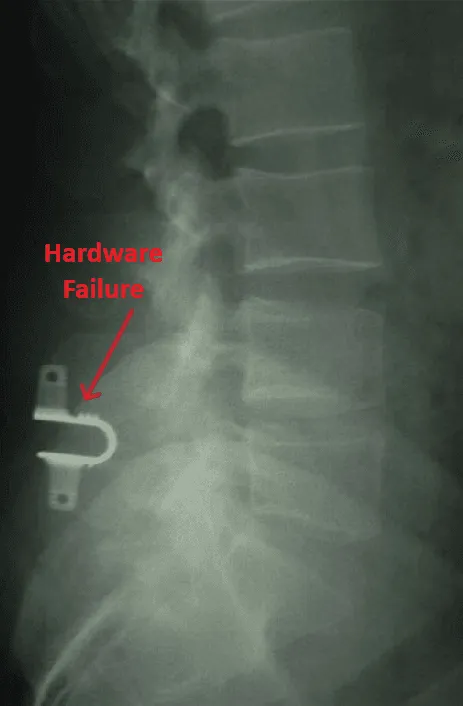
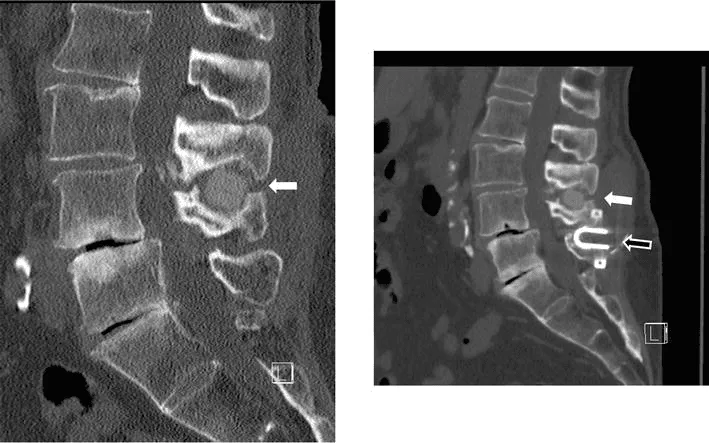
Implant migration or separation requiring emergency revision surgery.
Proximal junction kyphosis and failure causing abnormal spinal curvature above the device.
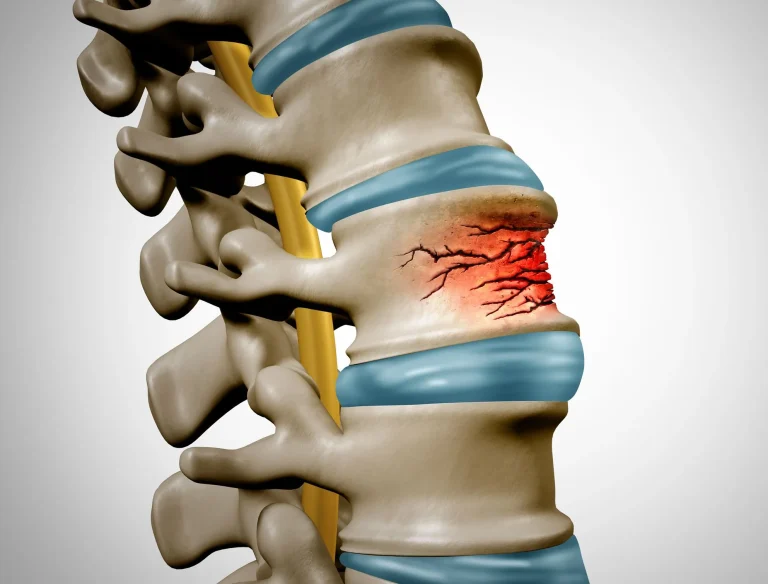
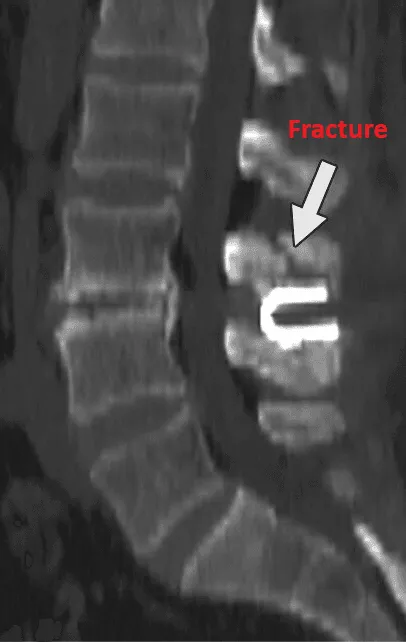
Vertebral fracture, subsidence, and telescoping of the implant into weakened bone.
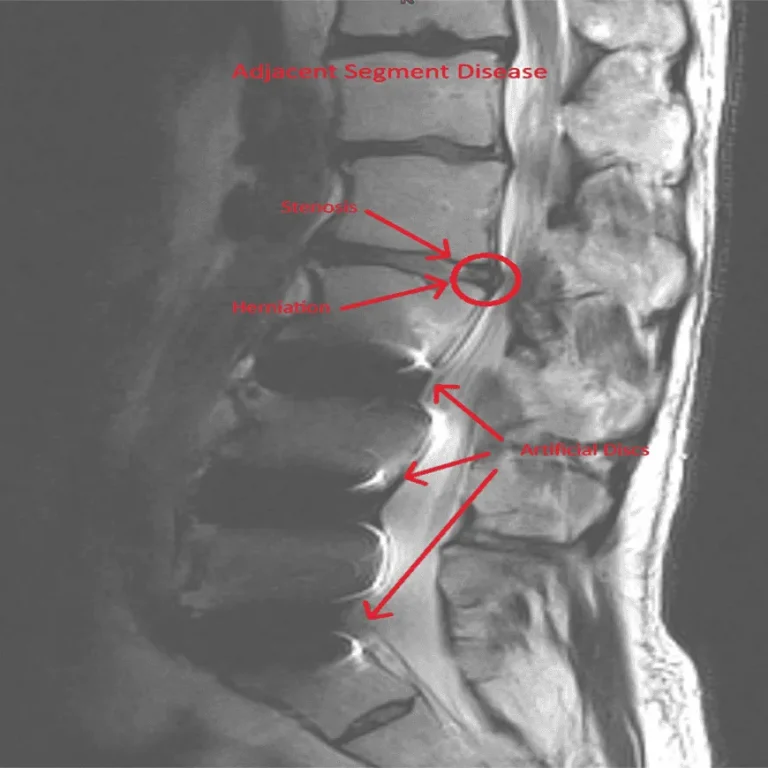
Adjacent segment disease and spinal instability caused by altered biomechanics.
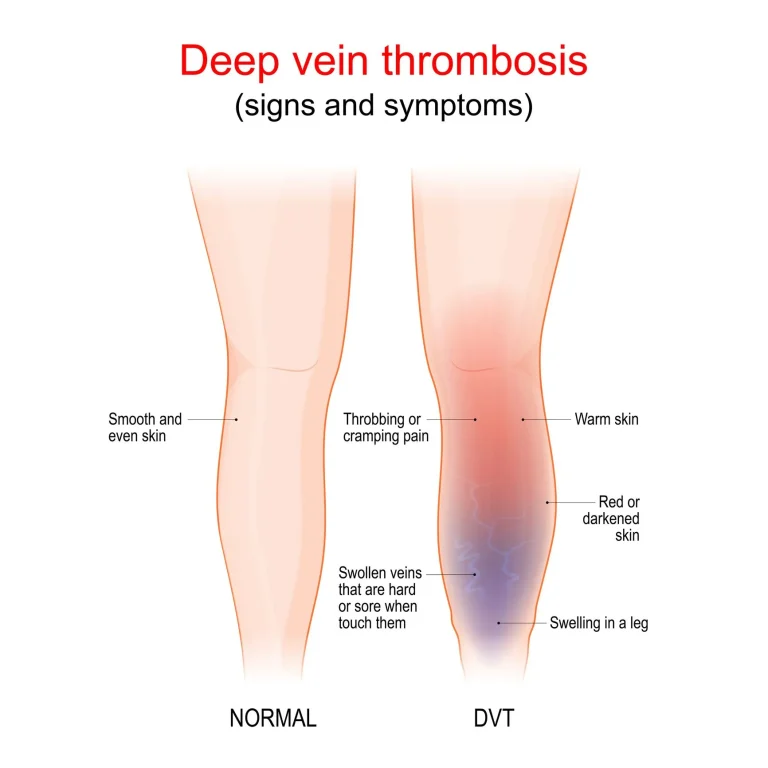
Deep venous thrombosis, pulmonary embolism, heart attack, and pneumothorax.
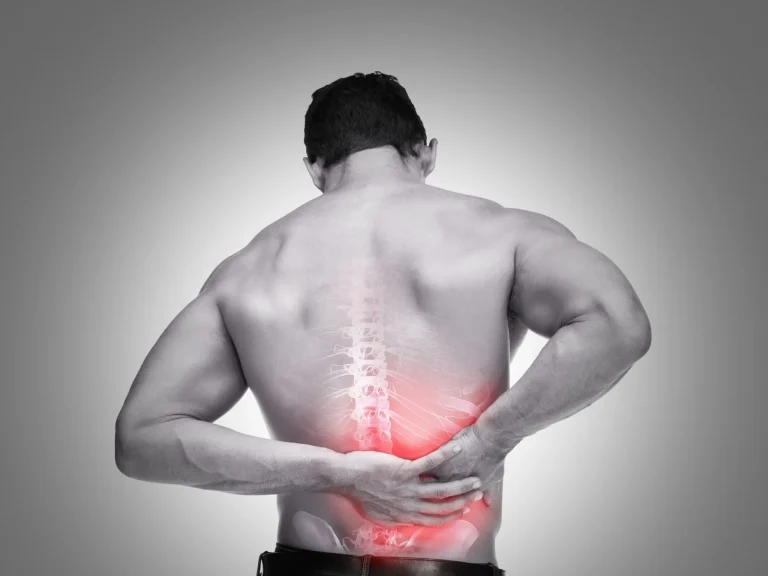
Permanent failed back surgery syndrome with unrelenting chronic pain.

Sexual dysfunction and retrograde ejaculation from surgical nerve damage.
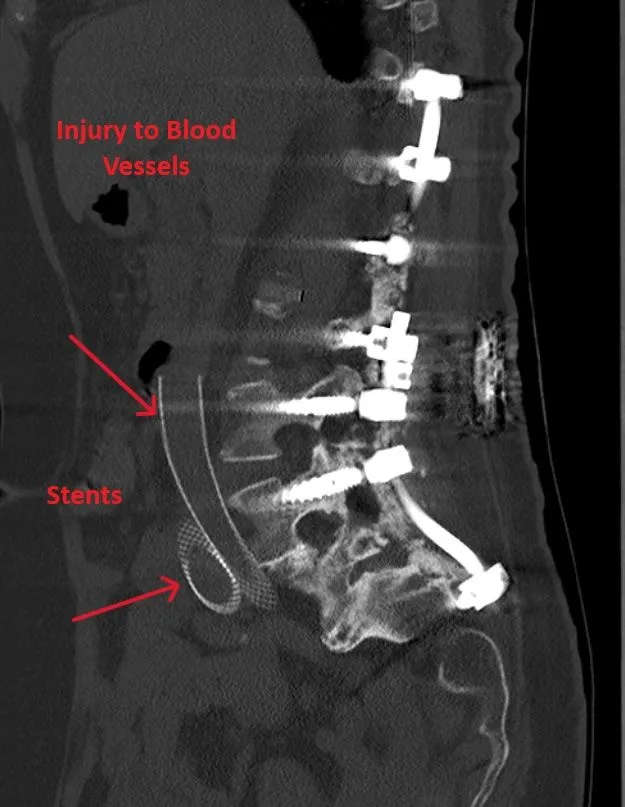
Injury to blood vessels, bladder, or ureters during surgical approach.
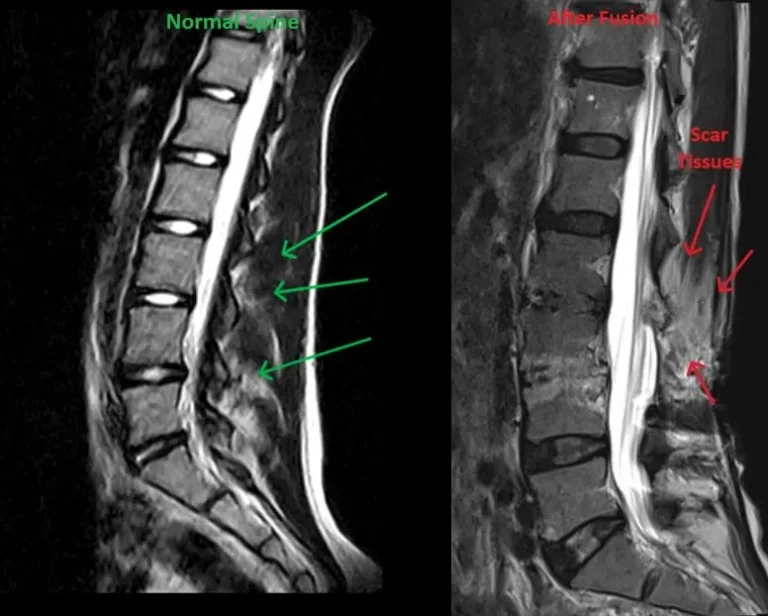
Excessive scar tissue formation around the spine and nerves post-surgery.
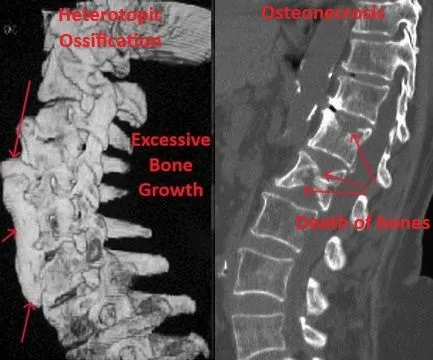
Heterotopic ossification and osteonecrosis — abnormal bone growth and bone death.

Fatal complications from surgery, anesthesia, blood clots, or post-operative events.
Excessive radiation exposure from fluoroscopy during device placement and follow-up imaging.
Allergic reactions to metal implant materials causing inflammation and tissue damage.
WHAT WE RECOMMEND INSTEAD
Instead of attaching metal hardware to your spine, Deuk Laser Disc Repair® uses endoscopic technology and laser precision to treat the actual source of pain — the damaged disc — through an incision smaller than a fingernail.
Your spine's stabilizing structures remain completely intact. No lamina, spinous process, or ligament destruction. No metal hardware attached.
Laser technology directly repairs the damaged disc — the actual source of pain that interspinous and interlaminar devices completely ignore.
Outpatient procedure under light sedation. Walk out the same day — no hospital stay, no intubation, no opioids required.
95% patient satisfaction, 0.01% complication rate, and over 1,300 successful procedures performed by Dr. Deukmedjian.
Feature
Interspinous/Interlaminar Devices
Deuk Spine Procedure
Procedure Type
❌ Invasive, hardware implantation
✅ Minimally invasive, endoscopic
Incision Size
❌ 2–4 inches
✅ Less than 1/4 inch
Anesthesia
❌ General (intubated)
✅ Light IV sedation
Hardware
❌ Metal implant attached to spine
✅ None
Treats Disc Injury
❌ No — disc left untreated
✅ Yes — laser repairs the disc
Hospital Stay
❌ 1–3 days inpatient
✅ Outpatient — go home same day
Recovery Time
❌ 4–8 weeks
✅ Days
Complication Rate
❌ High — device failure common
✅ 0.01%
Success Rate
❌ Variable — many end in pain management
✅ 95% patient satisfaction
Repeat Surgery
❌ Common — device migration, failure
✅ Rarely needed
A BETTER ALTERNATIVE
Minimally invasive, outpatient procedure with a 0.01% complication rate and 95% patient satisfaction. No bone removal. No metal hardware. Same-day recovery.
Patient Satisfaction
Complication Rate
Procedure Time