Procedure Not Recommended
Risks, Complications & Better Alternatives
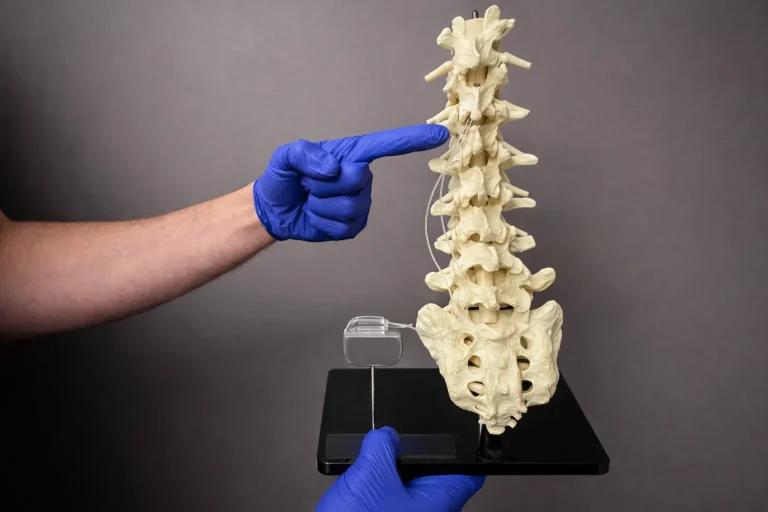
⚠️ While Deuk Spine Institute can perform Spinal Cord Stimulator implantation, we do not recommend it.
This page explains why — learn the risks before consenting to this procedure.
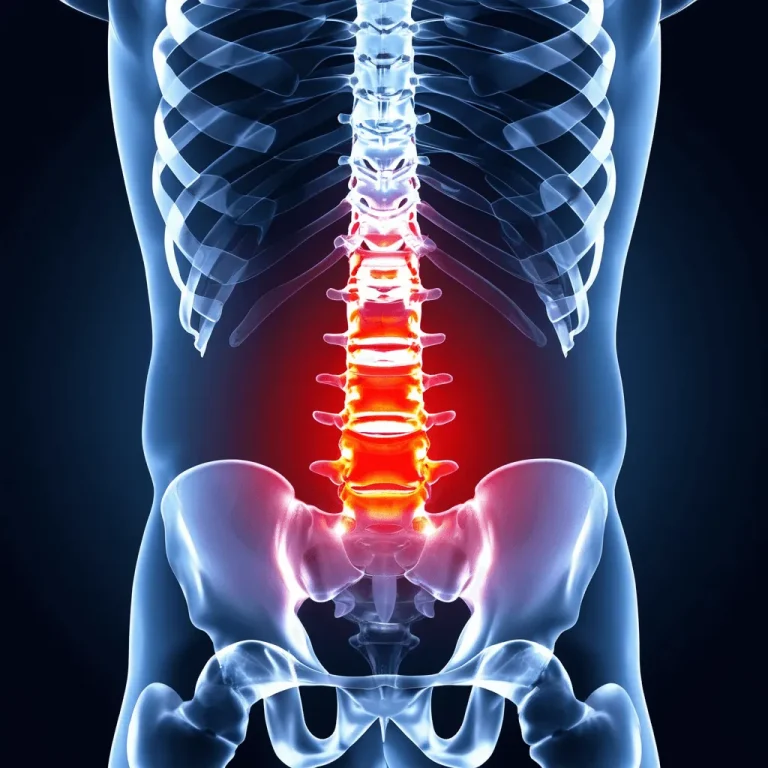
UNDERSTANDING THE PROCEDURE
While there are legitimate medical reasons, financial incentives often influence recommendations for this device-dependent procedure.
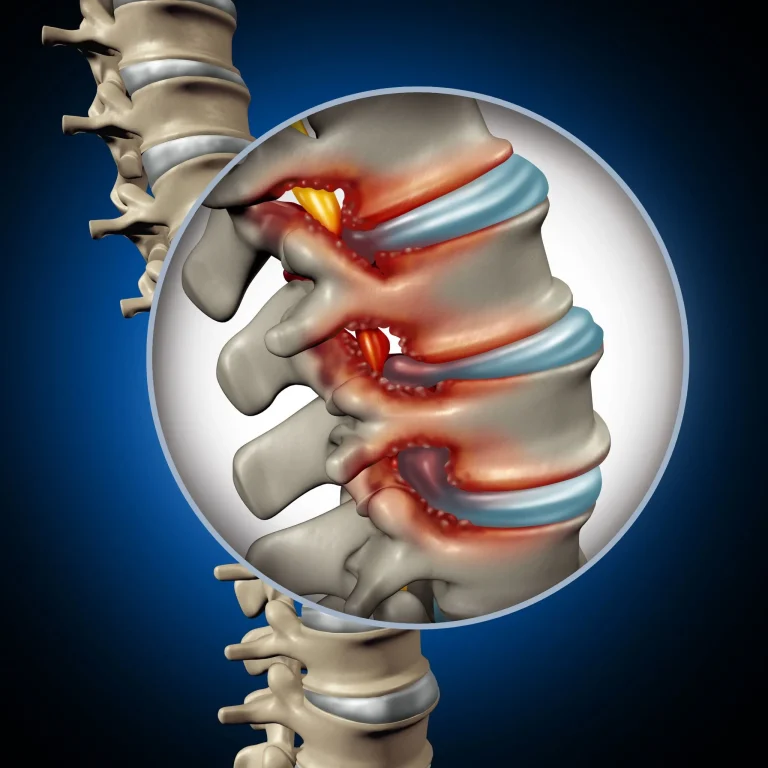
Nerve root dysfunction from compressed or pinched nerves. However, SCS masks the pain signal rather than treating the nerve compression itself.
Pain radiating down the leg to the foot from compressed nerves. SCS does not address the herniated disc or nerve compression causing the sciatic pain.
Poly-radiculopathy from compressed nerves. A rare emergency condition sometimes used to justify SCS for far less severe cases.
Pain from compressed or pinched spinal cord. SCS only masks the pain signal without addressing the structural damage causing compression.
General chronic back pain. SCS is often recommended as a last resort, but it never treats the source of pain — only masks it with electrical signals.
Pain management doctors are crossing into surgical territory to recommend and perform SCS implantation, often for excessive financial rewards with little benefit to the patient.
Among the most profitable procedures per case. Surgeon profits can exceed $10,000 per SCS implantation, creating powerful financial incentive.
The expensive implant device, multiple visits for programming and adjustments, trial period, and surgical facility fees drive massive hospital and device manufacturer revenue.
Battery replacements every 5-10 years, lead revisions, reprogramming visits, and device upgrades create a stream of recurring revenue for the lifetime of the patient.
Doctors receive the same compensation whether pain resolves or worsens. The system rewards implantation volume, not patient outcomes or pain resolution.
THE SURGICAL PROCESS
The videos below contain real surgical footage. Viewer discretion is advised.
Two separate incisions are made — one in the spine for lead placement and another in the buttock/abdomen for the generator.
The tough connective tissue covering the spine muscles is cut and removed to access deeper structures.
Spinal muscles are stripped from the bone, causing permanent damage and weakness to the stabilizing musculature.
The bony protrusion and stabilizing ligaments at the back of each vertebra are cut away with surgical instruments.
The lamina (protective bone arch) and ligamentum flavum are removed, exposing the spinal cord.
Facet joints that provide spinal stability are destroyed, often leading to instability requiring fusion.
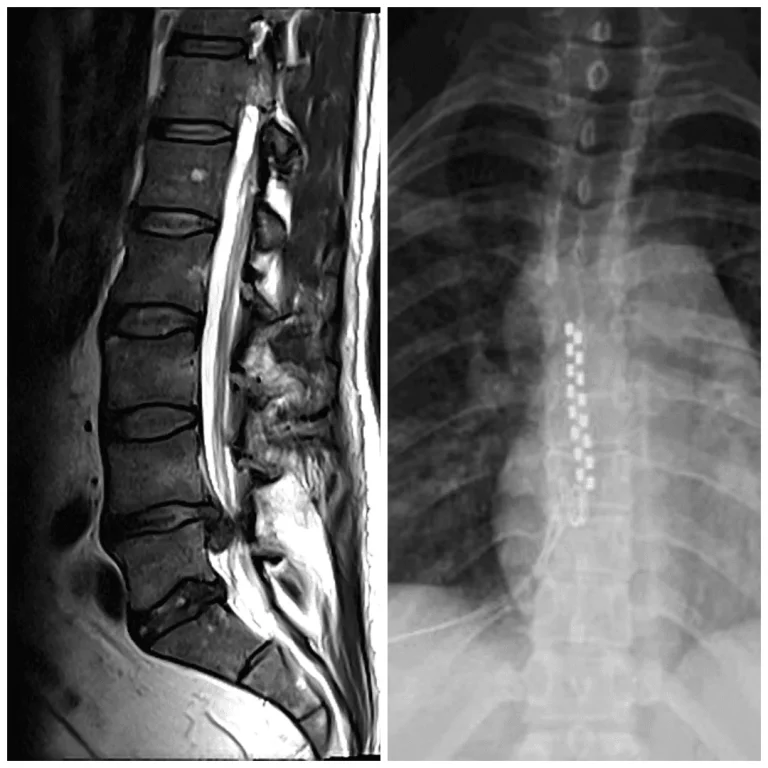
Electrode leads are carefully positioned directly on the spinal cord to deliver electrical stimulation signals.
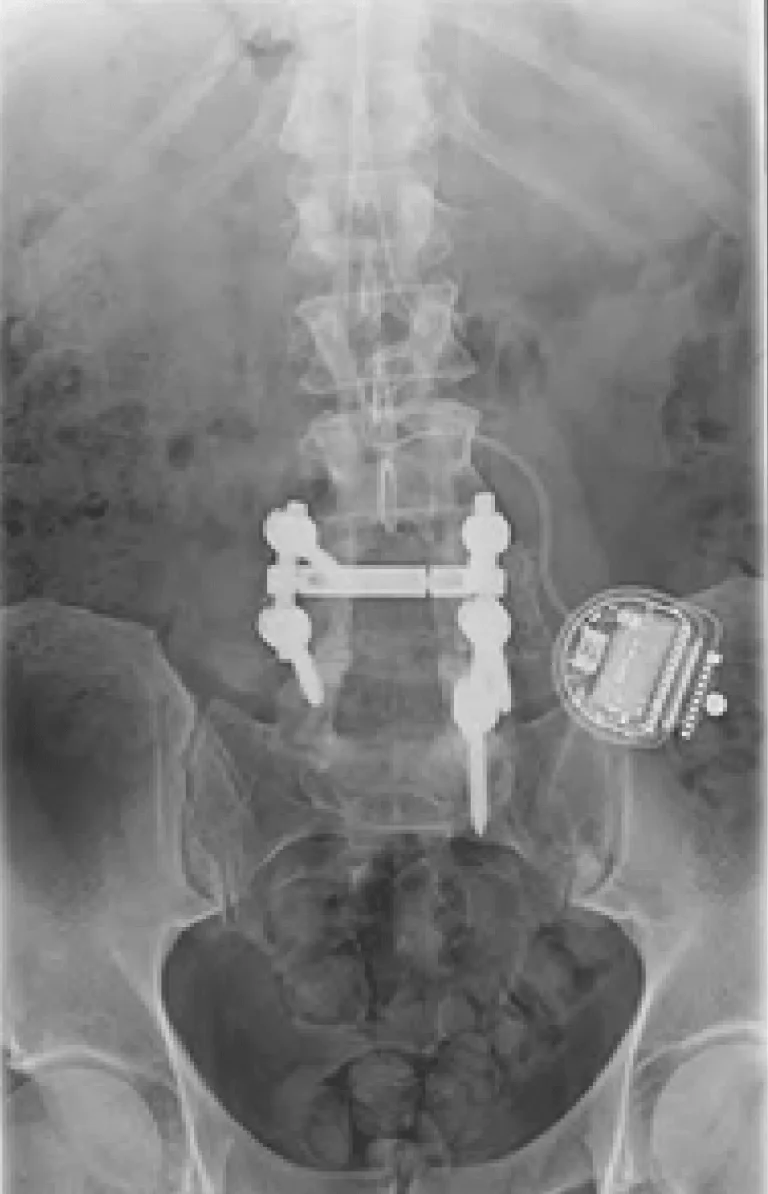
The leads are tunneled under the skin and connected to a pulse generator (battery) implanted in the buttock or abdomen.
CRITICAL RISKS
20 documented reasons with supporting images and surgical footage.
Images and videos below contain real surgical procedures and medical imagery.
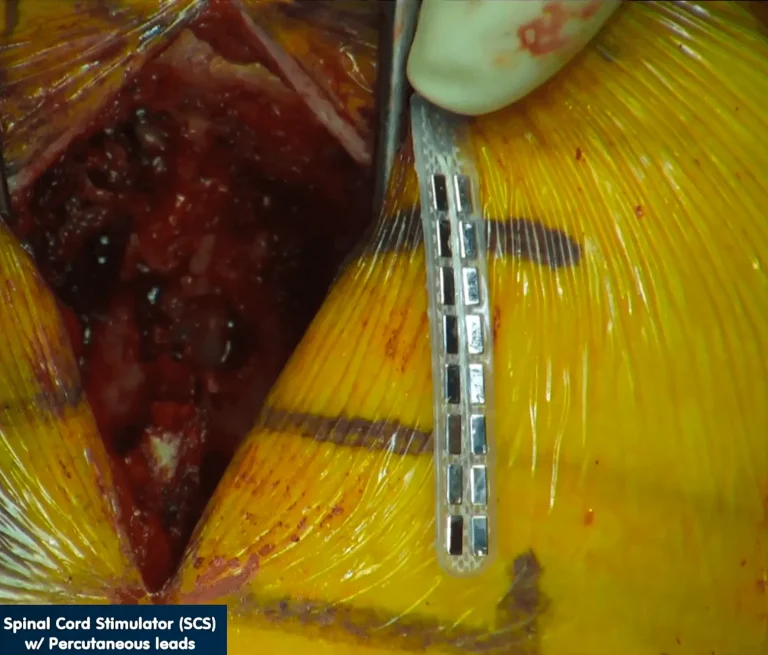
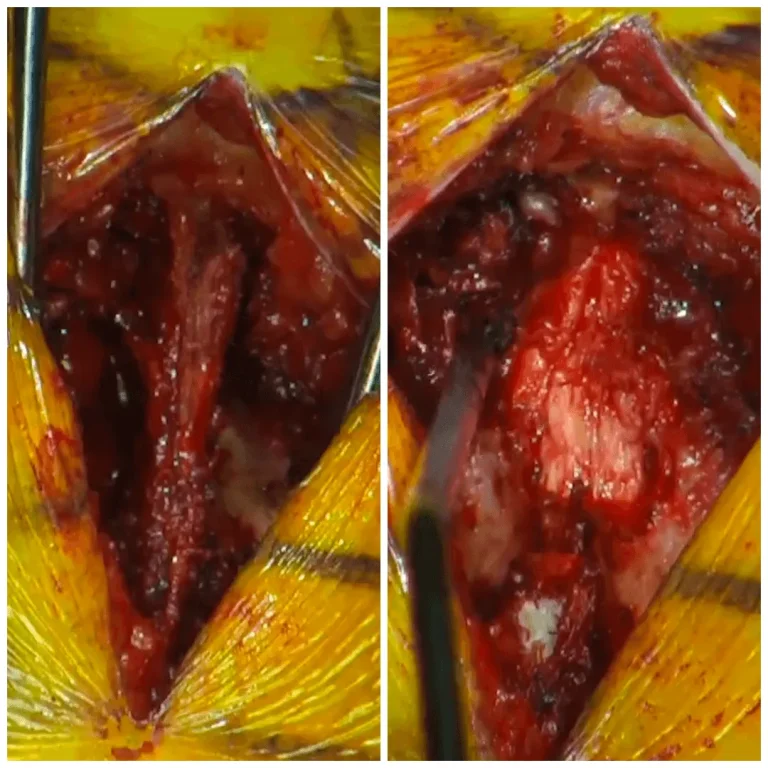
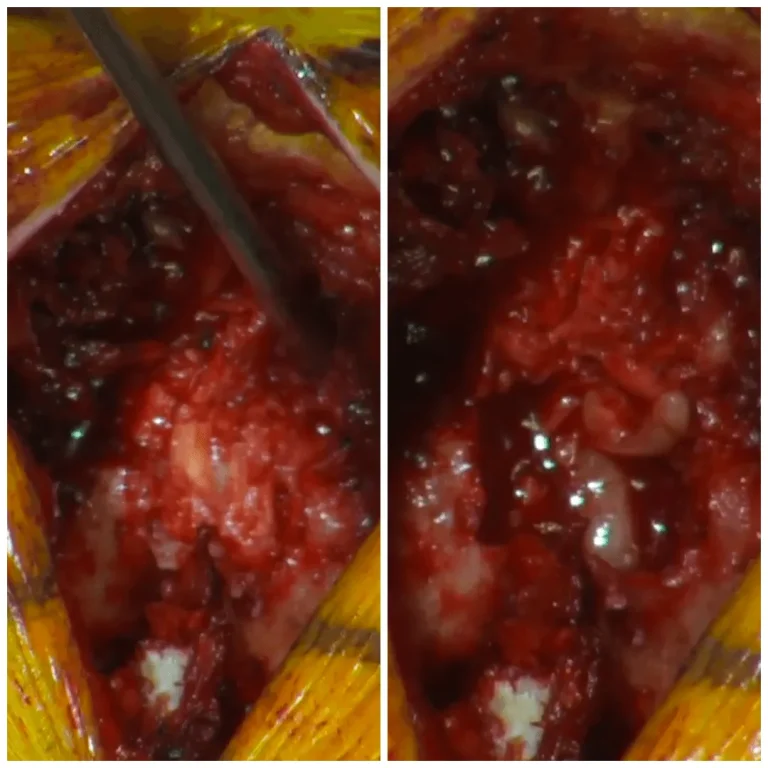
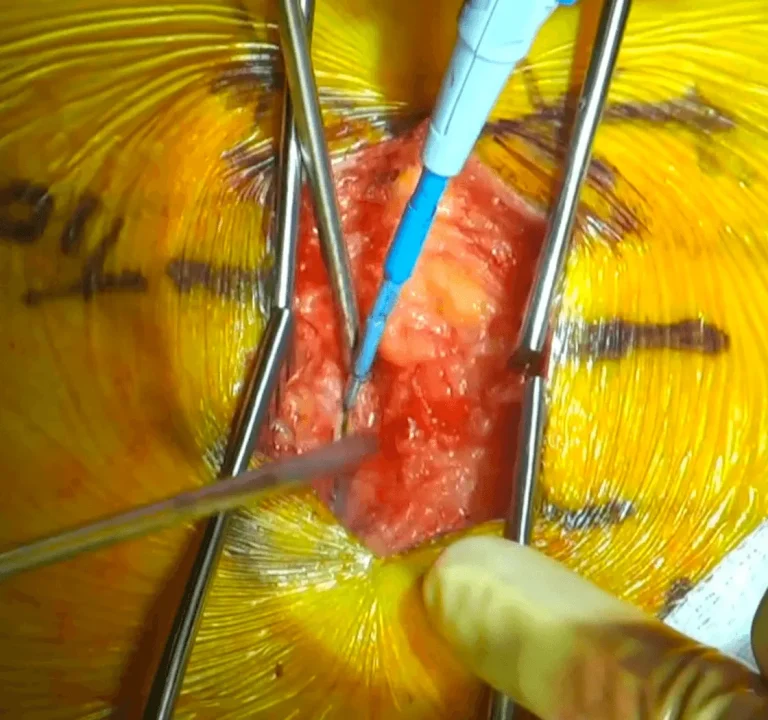
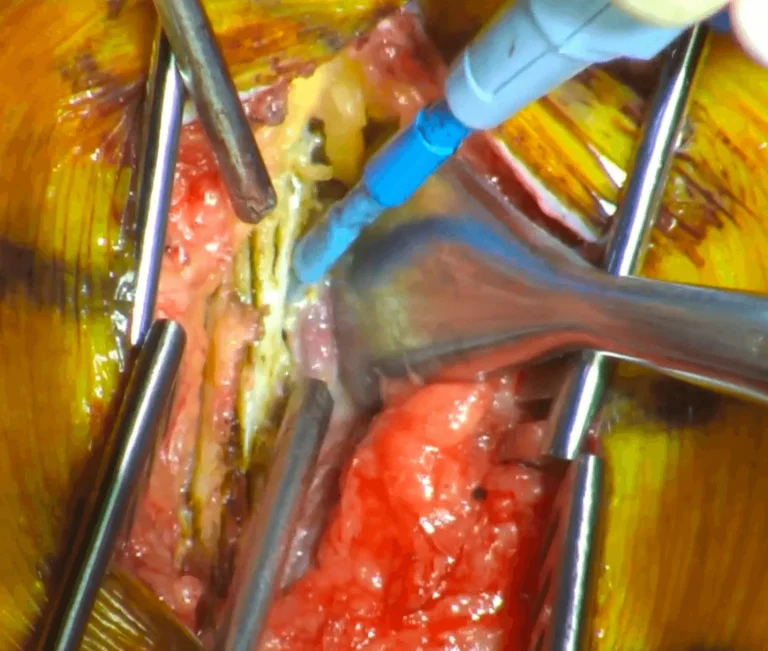
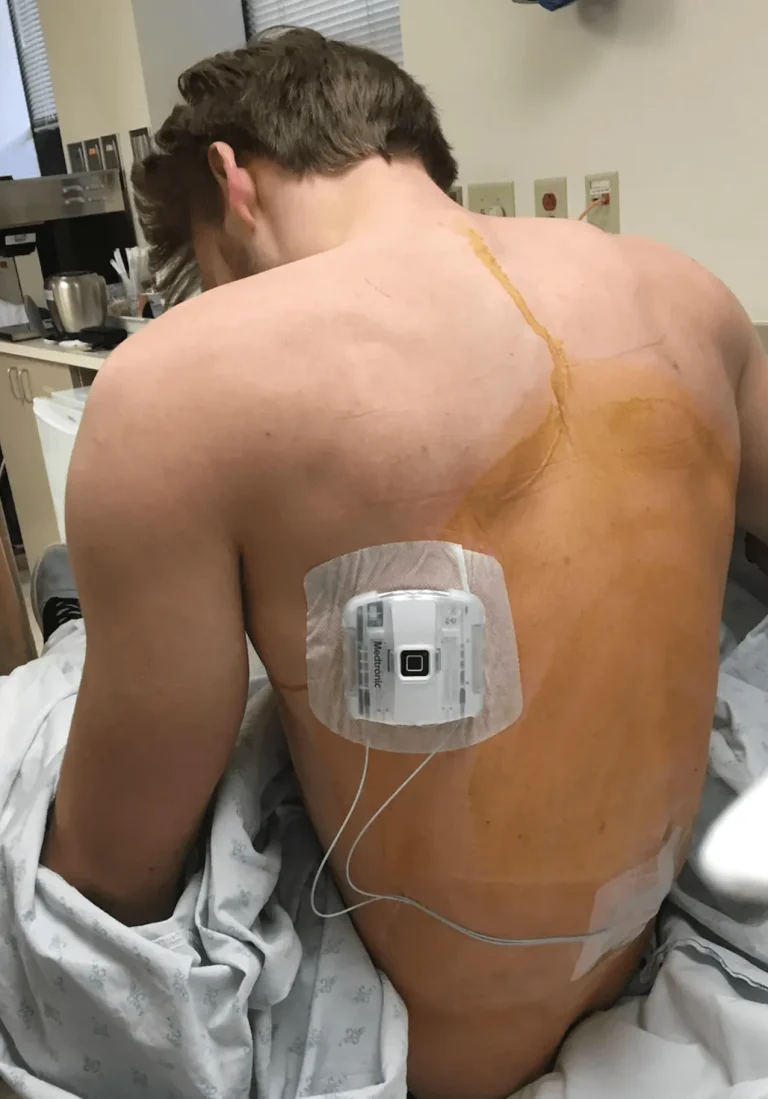
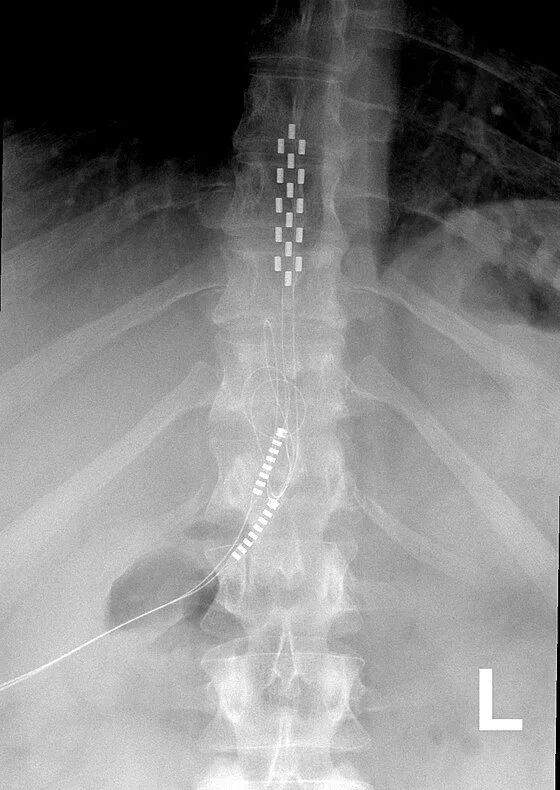
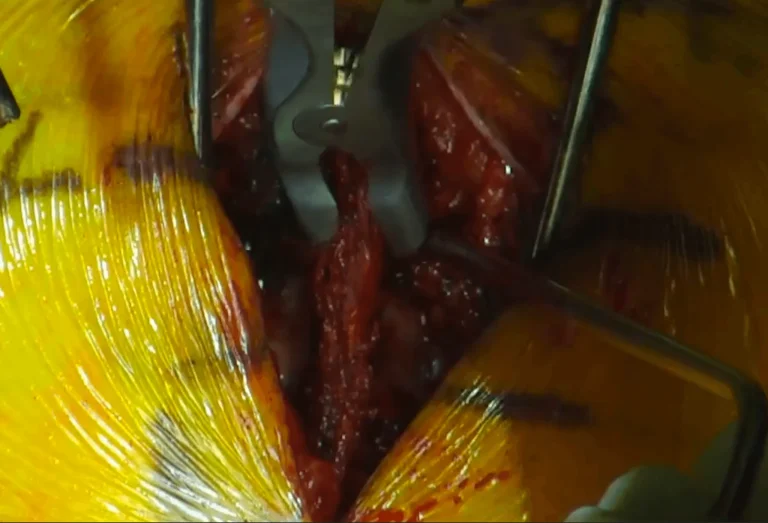
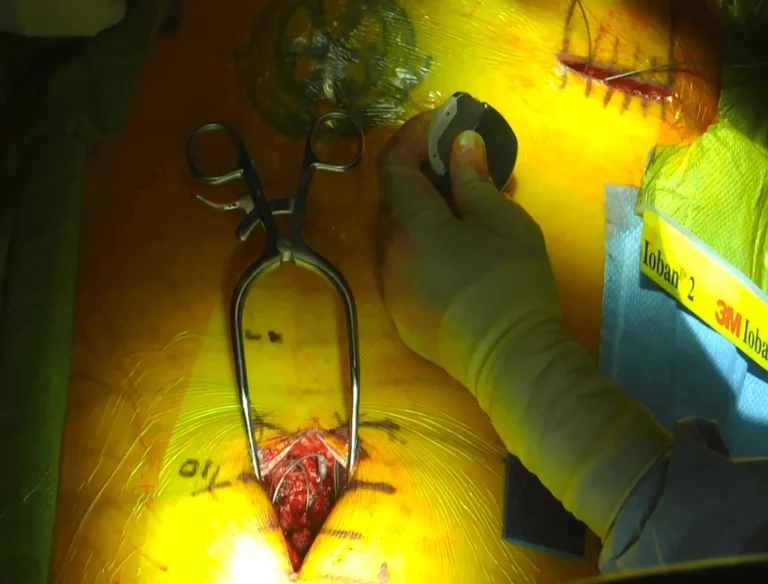
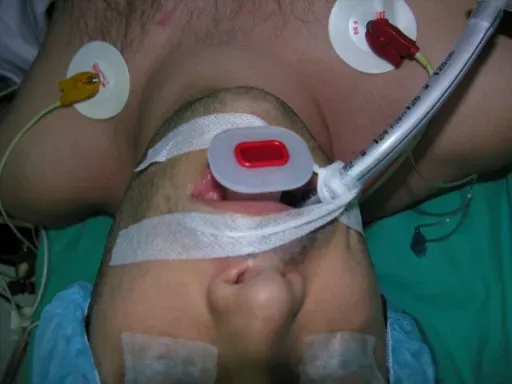
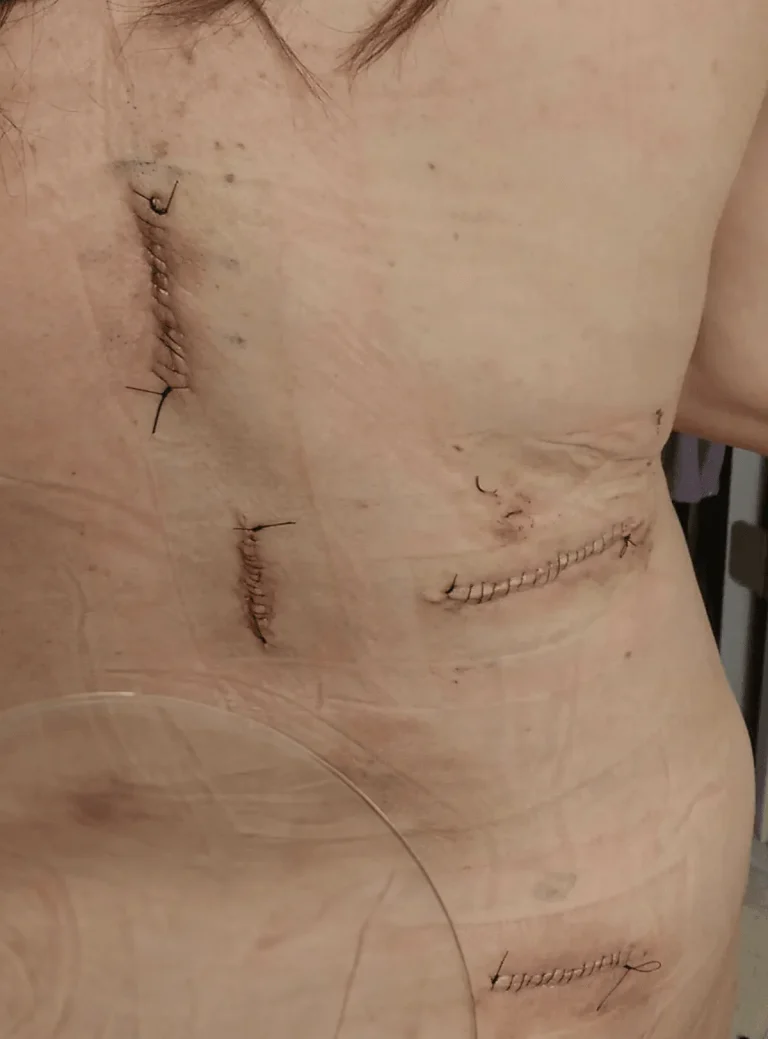
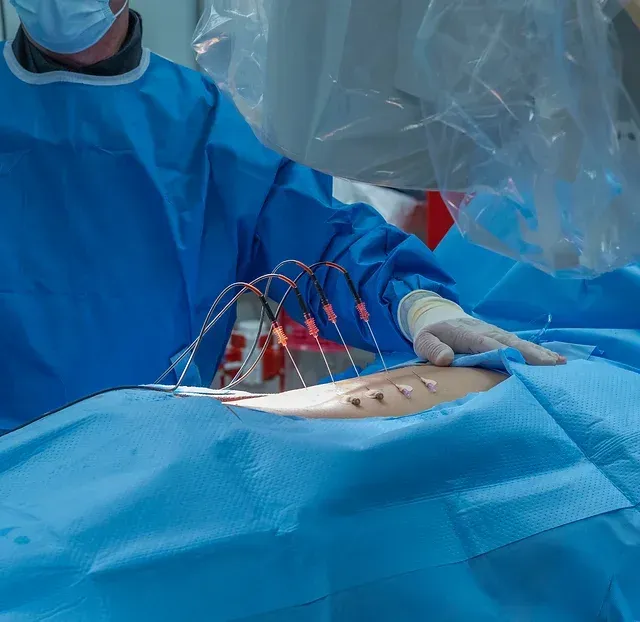
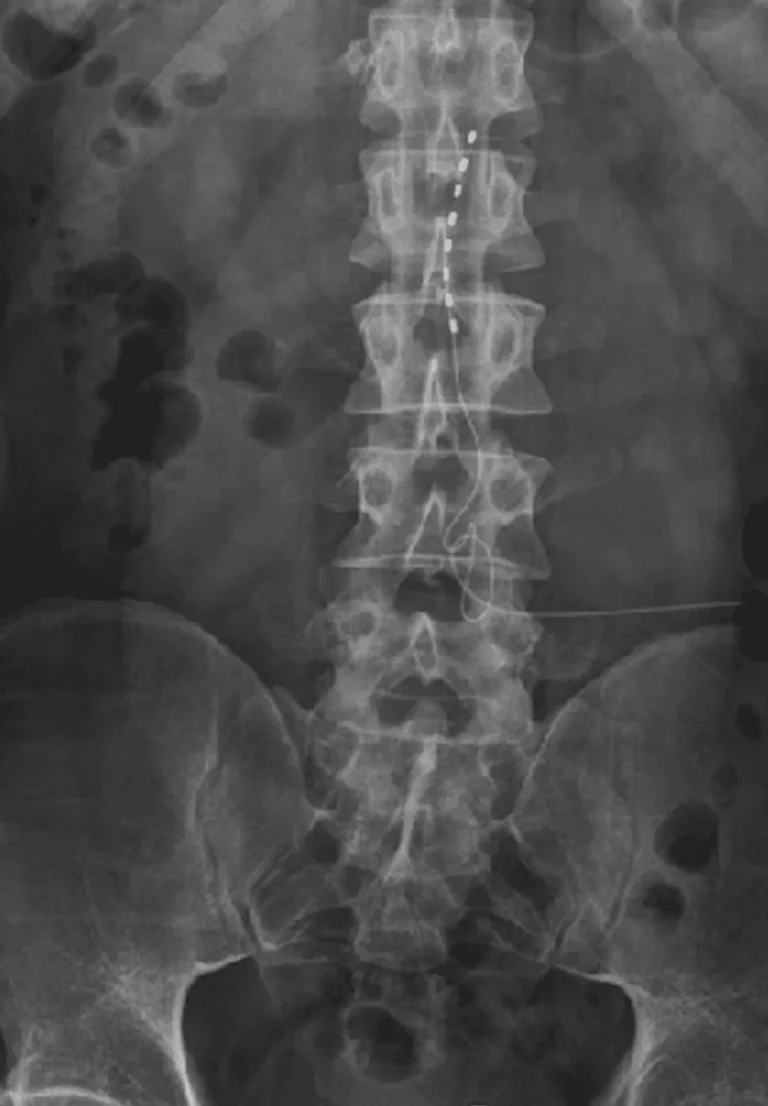
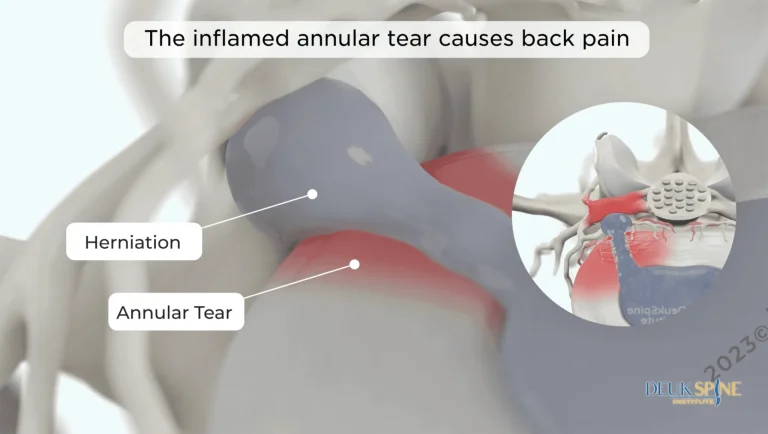
DOCUMENTED COMPLICATIONS
Images below show real surgical complications. Viewer discretion is advised.
Permanent nerve or spinal cord damage causing weakness, numbness, or paralysis in extremities.
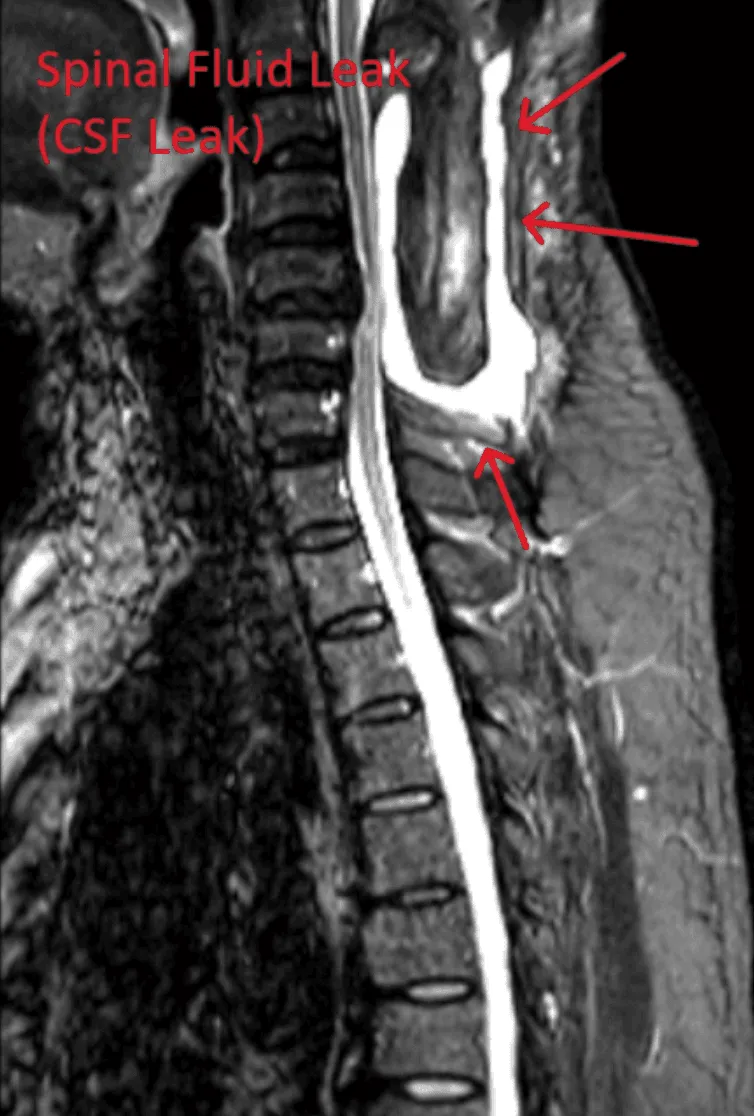
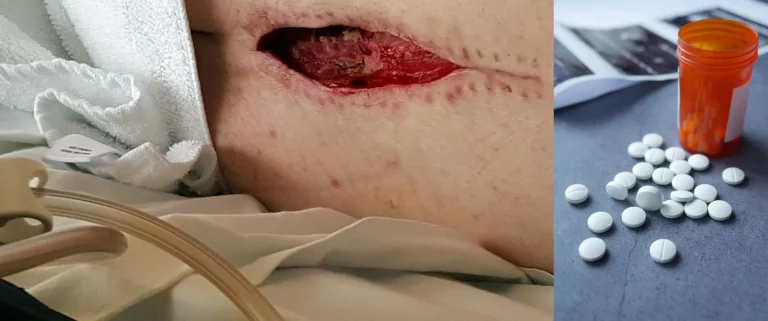
Dural tears during lead placement leading to cerebrospinal fluid leaks, requiring additional repair surgery.
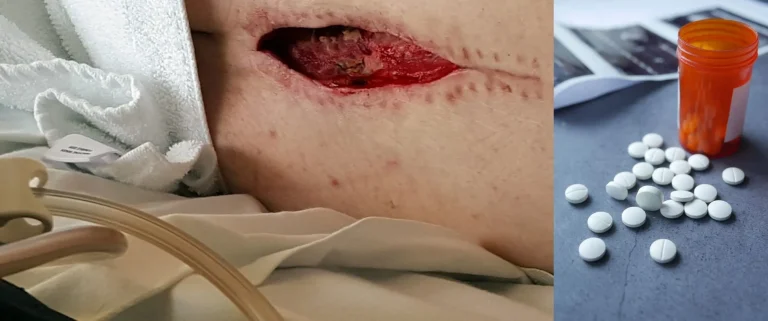
Including meningitis, spinal epidural abscess, osteomyelitis, and discitis from implanted hardware.
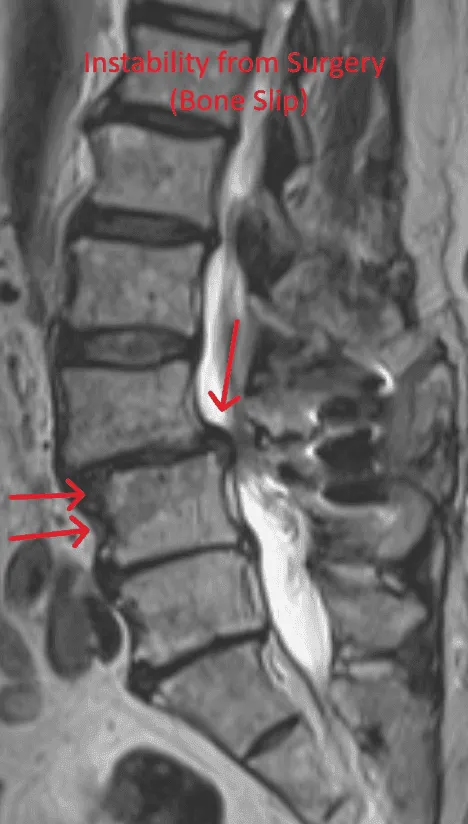
Laminectomy during placement removes stabilizing structures, causing instability requiring fusion with screws and rods.
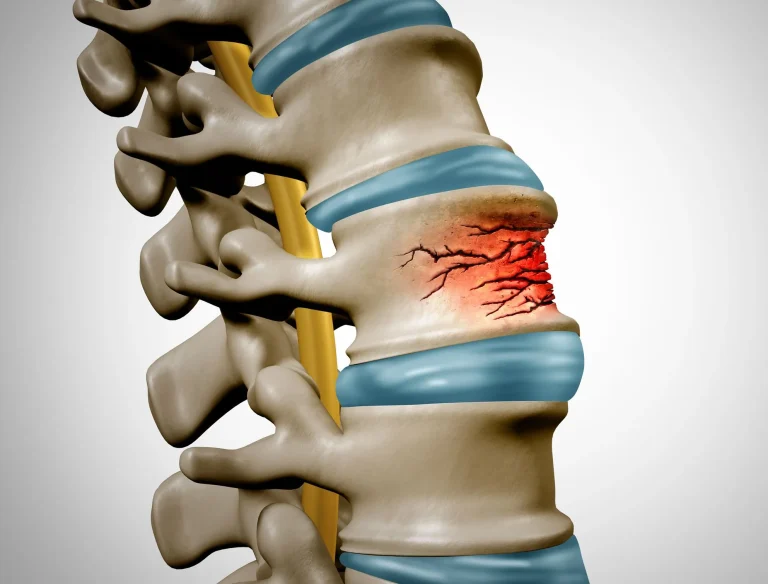
Weakened spine from bone removal during placement can lead to vertebral fractures and collapse.
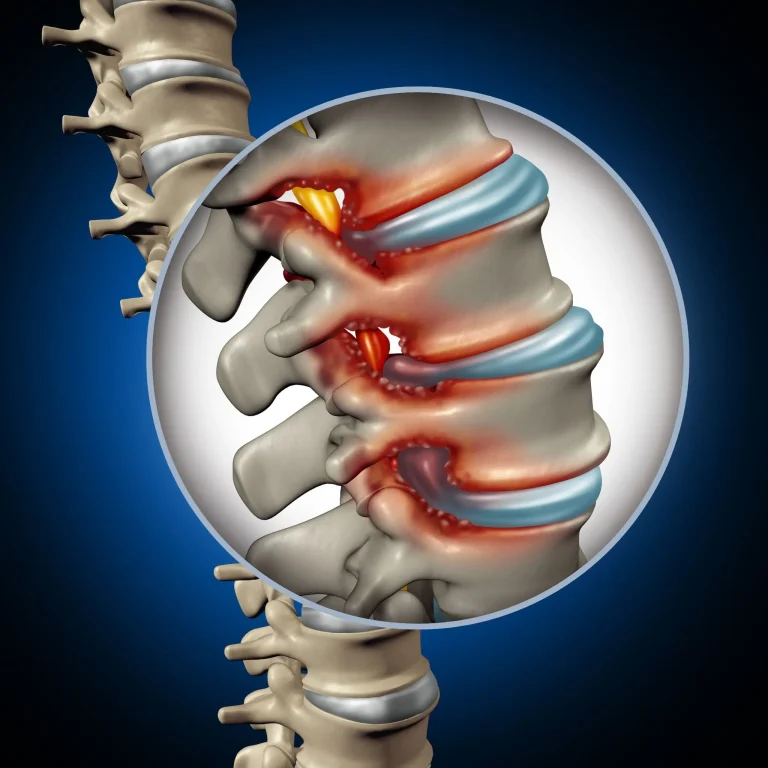
Stenosis persists or reoccurs after SCS placement, requiring additional surgical intervention.
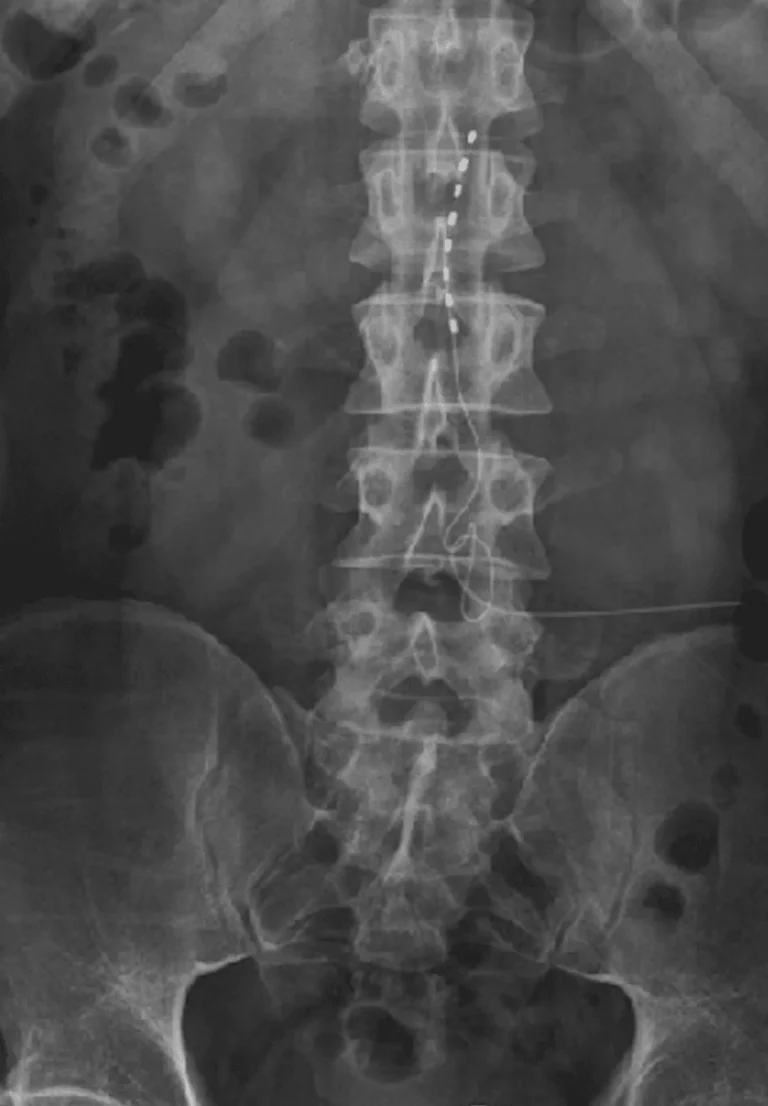
Implanted leads shift from their original position, causing loss of pain relief and requiring revision surgery.
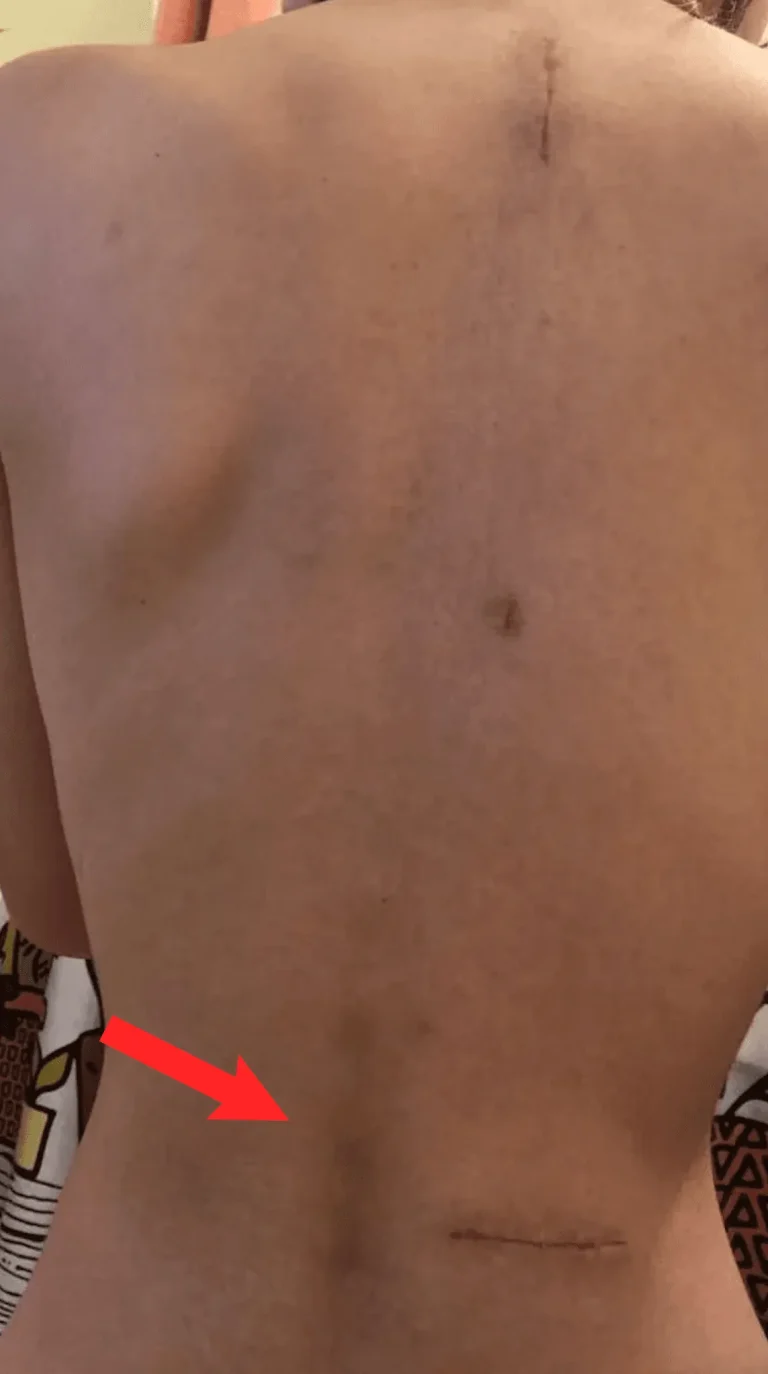
Excessive scar tissue forms around the spinal cord and nerves, causing chronic pain and nerve dysfunction.

Lead placement on the spinal cord carries risk of permanent paralysis from cord damage during surgery.
WHAT WE RECOMMEND INSTEAD
Instead of masking pain with electrical signals, Deuk Laser Disc Repair® uses endoscopic technology and laser precision to treat the actual source of pain — the damaged disc — through an incision smaller than a fingernail. For piriformis-related pain, Deuk Piriformis Release offers targeted relief, and Deuk Plasma Rhizotomy® addresses facet joint pain without hardware or device dependency.
Treats the source of pain directly — no permanent implant, no leads, no generator, no battery replacements for life.
Laser technology directly repairs the damaged disc — the actual source of pain that SCS merely masks with electrical signals.
Outpatient procedure under light sedation. Walk out the same day — no hospital stay, no intubation, no opioids required.
95% patient satisfaction, 0.01% complication rate, and over 1,300 successful procedures performed by Dr. Deukmedjian.
Feature
Spinal Cord Stimulator
Deuk Laser Disc Repair®
Approach
❌ Masks pain with electrical signals
✅ Treats the source of pain
Procedure
❌ Invasive — requires laminectomy
✅ Minimally invasive, endoscopic
Hardware
❌ Permanent implant (leads + generator)
✅ None
Incision Size
❌ Two large incisions
✅ Less than 1/4 inch
Anesthesia
❌ General (intubated)
✅ Light IV sedation
Hospital Stay
❌ 1-3 days inpatient
✅ Outpatient — go home same day
Recovery Time
❌ 4-8 weeks
✅ Days
Device Dependency
❌ Lifetime — battery replacements required
✅ None
Complication Rate
❌ High — lead migration, paralysis risk
✅ 0.01%
Success Rate
❌ Variable — many end in pain management
✅ 95% patient satisfaction
A BETTER ALTERNATIVE
Minimally invasive, outpatient procedure with a 0.01% complication rate and 95% patient satisfaction. No implants. No device dependency. Same-day recovery.
Patient Satisfaction
Complication Rate
Procedure Time